|
4/10/2023 0 Comments Solid particles movement 
With an increase in temperature, the particles move faster as they gain kinetic energy, resulting in increased collision rates and an increased rate of diffusion. In liquids, particles are quite close together and move with random motion throughout the container. Particles move rapidly in all directions but collide with each other more frequently than in gases due to shorter distances between particles. If the vertical motion of gas molecules did not slow under gravity, the atmosphere would have long since escaped from the Earth. Particles are still subject to gravity and hit the bottom of a container with greater force than the top, thus giving gases weight. It is the perfectly elastic nature of the collisions that enables the gas particles to continue rebounding after each collision with no loss of speed. This is very different to most other collisions where some kinetic energy is transformed into other forms such as heat and sound. Unlike collisions between macroscopic objects, collisions between particles are perfectly elastic with no loss of kinetic energy. The oxygen and nitrogen molecules in air at normal room temperature are moving rapidly at between 300 to 400 metres per second. The actual average speed of the particles depends on their mass as well as the temperature – heavier particles move more slowly than lighter ones at the same temperature. With an increase in temperature, the particles gain kinetic energy and move faster. In gases the particles move rapidly in all directions, frequently colliding with each other and the side of the container. We use multiple models of atoms to help explain chemical processes and describe their behaviour. 
Student 2: "When a block of ice is taken out of a freezer, the sudden change of temperature reacts on the particles making them decrease in size." Scientific viewĪtoms are incredibly small and cannot be seen with even the most powerful light microscope. When they have broken away from each other, they turn from a crystal form to a solution form." Student 1: “The particles start to break away from each other because of the rise in temperature. Some examples of students’ thinking about the behaviour of particles in a melting ice block are: Students frequently find it difficult to appreciate particle movement in solids and this leads to different conceptions about freezing and melting. When gases condensed to a liquid, many students attributed this to increased attractive forces between particles. Students stated that particles were forced apart (by heat acting as a substance) when gases were heated. When asked, “Why don't gas particles fall to the bottom of a vessel?” only about 50% of students thought that the particles were in constant motion. Random particle motion in liquids and gases is a difficult concept for students to appreciate. Students may believe that gas particles are moving slowly in ways similar to what they observe when they see suspended dust particles in a beam of light. Students frequently fail to understand the dynamic nature of particles they tend to think of them as static. These ideas are also explored in the focus idea 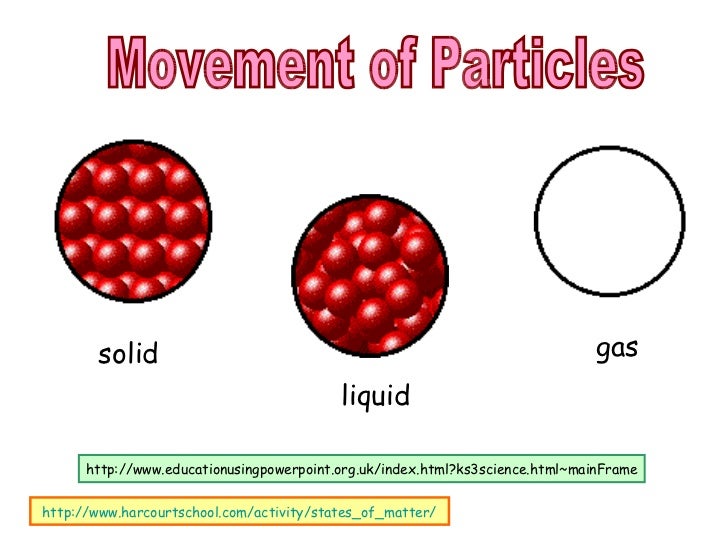
Students visualise atoms, molecules and ions to be little ball-like objects (perhaps because of the way the information has been presented) and this contributes to them confusing the properties of the particles with the macroscopic nature of the materials that they make up. Many students who appreciate that matter is particulate still retain some former views and consider that particles can change their form (solid to liquid), explode, burn, expand, change shape and colour or shrink. They often lack an appreciation of the very small size of particles, attribute macroscopic properties to microscopic particles, have difficulty appreciating the motion of particles in all states of matter and have problems understanding forces between particles. Research evidence suggests that many students at this age and older still hold a number of alternative conceptions about particles which prove difficult to extinguish. Contrasting student and scientific viewsĬontrasting student and scientific views Student everyday experiencesĪt this level, students are expected to 'explain the behaviour and properties of materials in terms of their constituent particles and the forces holding them together’ (VELS standards Level 6). However, the fact that students may be able to draw the usual static arrangements of particles in solids, liquids and gases does not mean that they hold a fully particulate view of matter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
